 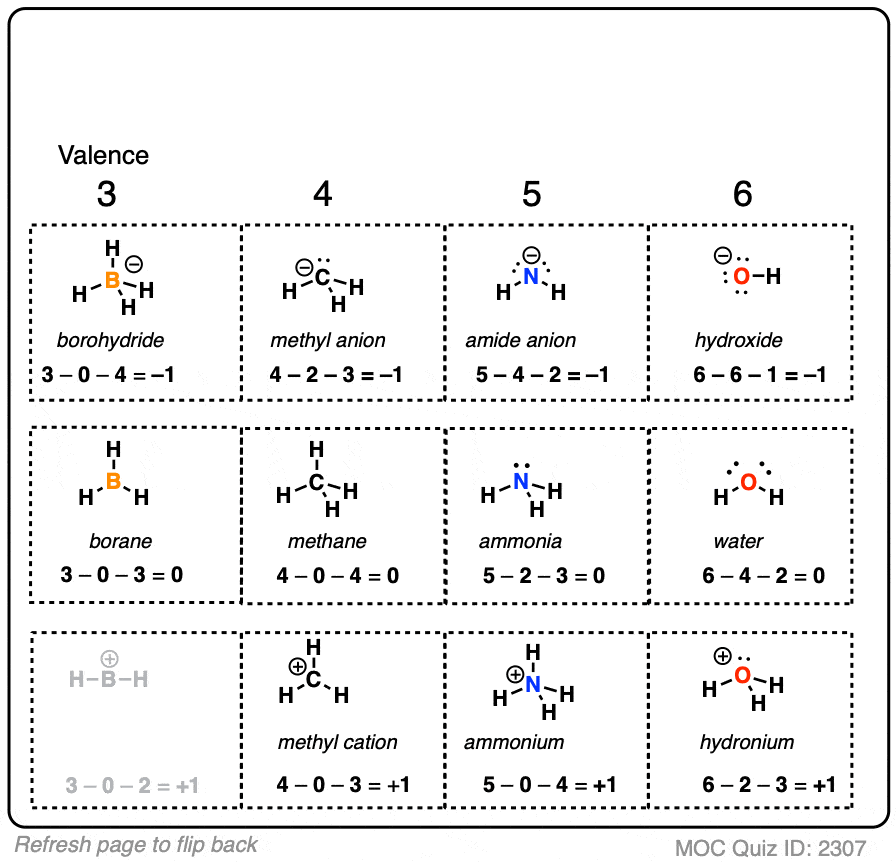
A negative formal charge indicates that an atom is more likely to be the source of electrons in a reaction (a nucleophile). The formal charge of a molecule can indicate how it will behave during a process. This condition could point to resonance structures, especially if the structures have the same atom arrangement but different types of arrangements of bonds. While formal charge can indicate a molecule's preferred structure, the problem becomes more complicated when numerous equally preferred structures exist. If there are numerous alternatives for a molecule's structure, this gives us a hint: the one with the least/lowest formal charges is the ideal structure. In BH4, the formal charge of hydrogen is 1-(0+1), resulting in a formal charge of 0.Ĭalculate the formal charge on the following:Īns: We are showing how to find a formal charge of the species mentioned.įormal charge on Cl atom of HClO4 ion: 7 – 8/2 – 0 = 3įormal charge on S atom of HSO4- ion: 6 – 8/2 – 0 = 2Īn atom in a molecule should have a formal charge of zero to have the lowest energy and hence the most stable state. One valence electron, zero non-bonded electrons, and one bond make up hydrogen. Let us now examine the hydrogen atoms in BH4. This changes the formula to 3-(0+4), yielding a result of -1. Take the compound BH 4 or tetrahydrdoborate.īoron (B) possesses three valence electrons, zero non-bonded electrons, and four bonds around it. The number of bonding electrons divided by two equals the number of bonds that surround the atom, hence this expression can be reduced to:įormal Charge = (number of valence electrons in neutral atom)-(non-bonded electrons + number of bonds) (Number of valency electrons in neutral atom)-(electrons in lone pairs + 1/2 the number of bonding electrons) The formula for computing a formal charge is: L = Number of non-bonding valence electrons on this atom in the moleculeī = Total number of electrons shared in bonds with other atoms in the moleculeįormula, Calculation, Importance, and Example V = Valence Electron of the neutral atom in isolation The following equation can be used to compute the formal charge of an atom in a molecule: 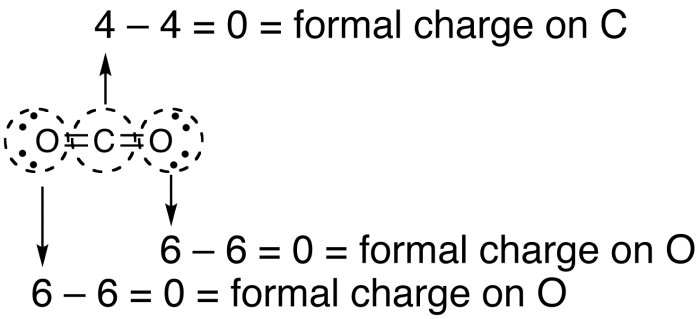
When choosing the optimum Lewis structure (or predominant resonance structure) for a molecule, it is important to keep the formal charge on each of the atoms as low as feasible. 
The formal charge is the difference between an atom's number of valence electrons in its neutral free state and the number allocated to that atom in a Lewis structure. or q) is the charge assigned to an atom in a molecule in the covalent view of bonding, assuming that electrons in all chemical bonds are shared equally between atoms, regardless of relative electronegativity. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
